![PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39bb0190c68df2ae004d05d1f71f4cce7460deed/2-Table1-1.png)
PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar
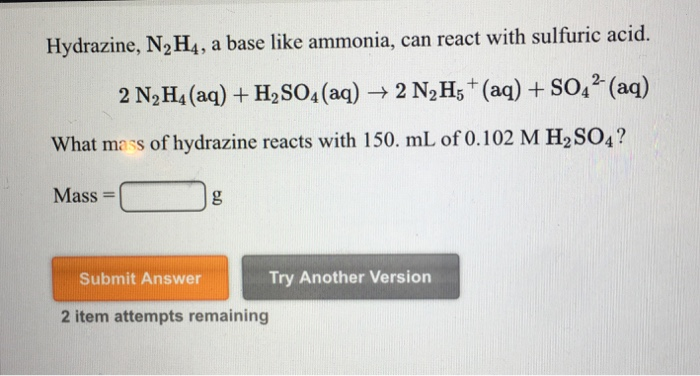
✓ Solved: Hydrazine, N2H4, is a weak base and can react with an acid such as sulfuric acid: 2N2H4(aq)+H2SO4(aq)→...

Electrochemical Strategy for Hydrazine Synthesis: Development and Overpotential Analysis of Methods for Oxidative N–N Coupling of an Ammonia Surrogate | Journal of the American Chemical Society

AMPP Store - 51318-11012-Amine base Vapor Phase Corrosion inhibitor Alternatives to Hydrazine for Steam generating system and
Hydrazine, diamine, diazane, N2H4 molecule. It is highly reactive base and reducing agent. Molecular model. 3D rendering. Illustration Stock Photo - Alamy
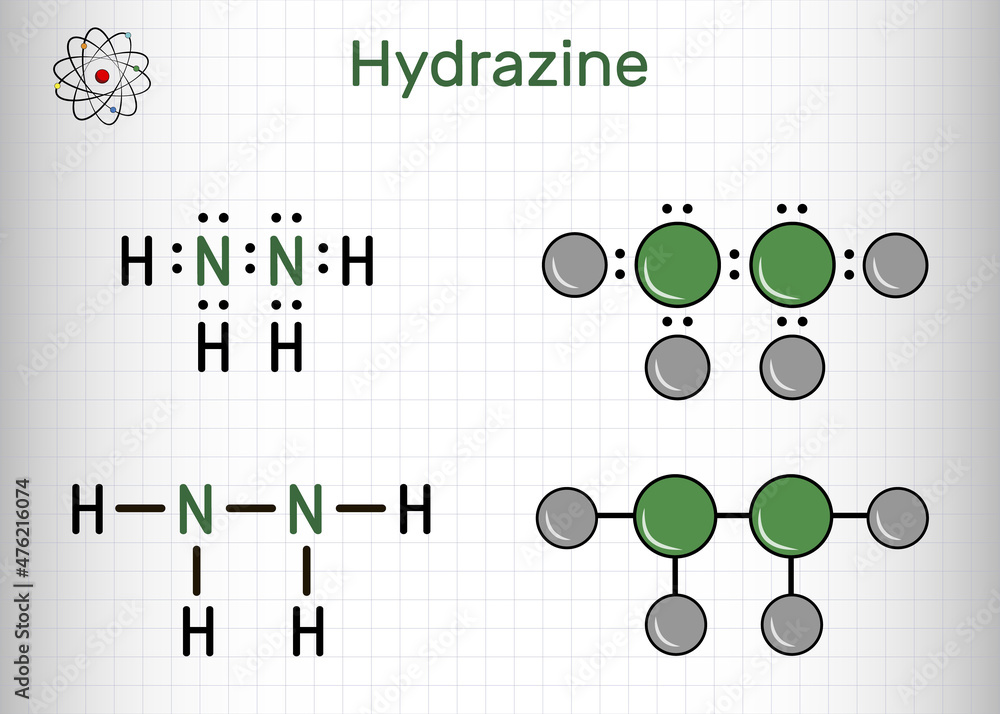
Hydrazine, diamine, diazane, N2H4 molecule. It is highly reactive base and reducing agent. Structural chemical formula and molecule model. Sheet of paper in a cage Stock Vector | Adobe Stock

China Reasonable price Hydrazine Base - Polyethylene Glycol – YANXA factory and manufacturers | YANXA
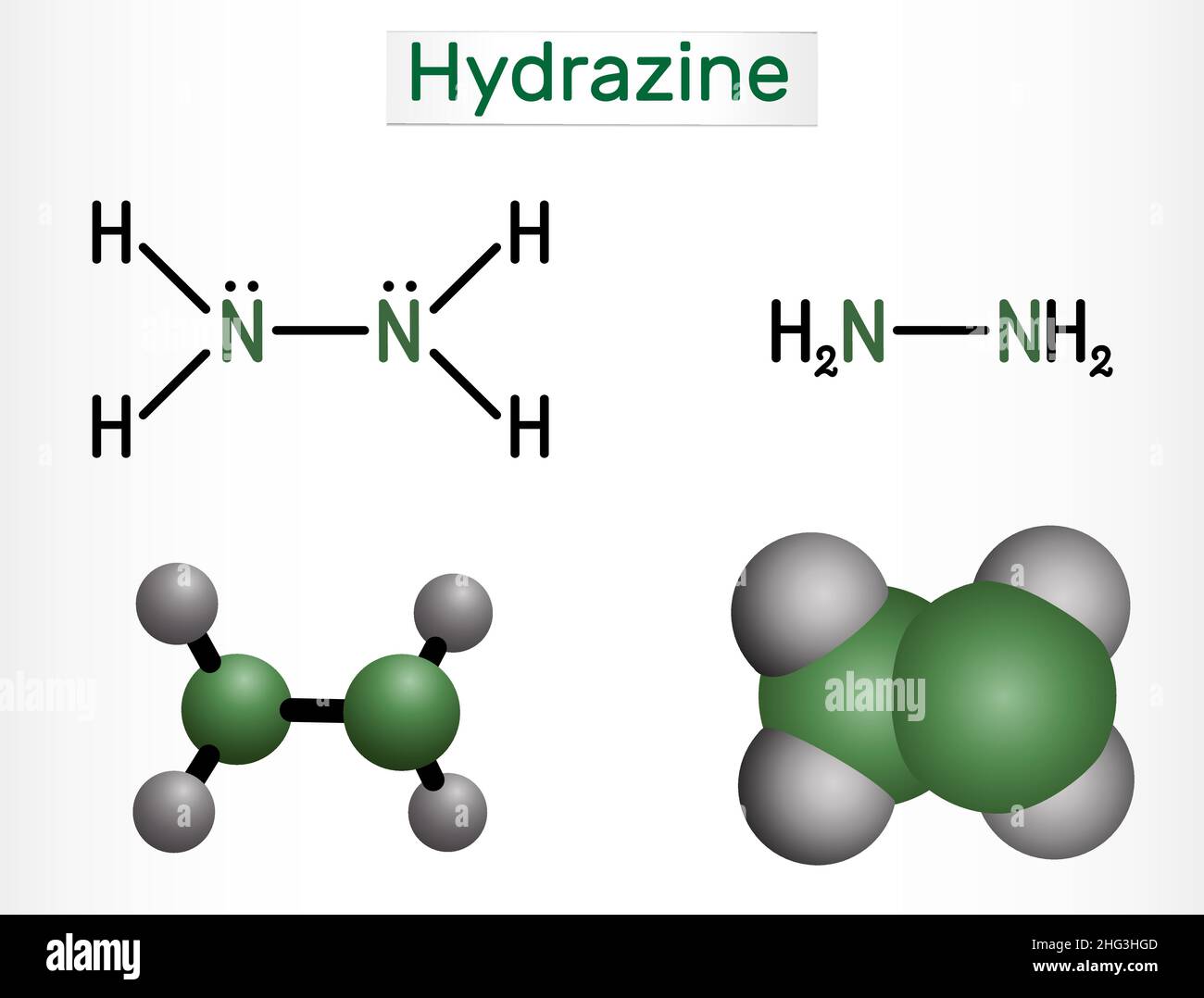
Hydrazine, diamine, diazane, N2H4 molecule. It is highly reactive base and reducing agent. Structural chemical formula and molecule model. Vector illu Stock Vector Image & Art - Alamy




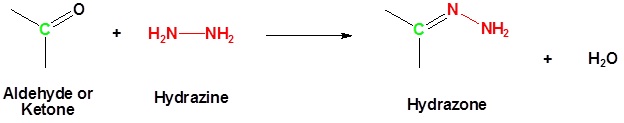



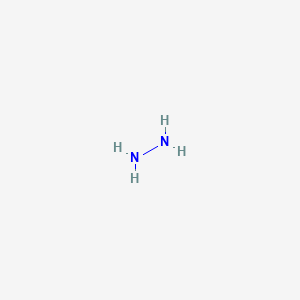



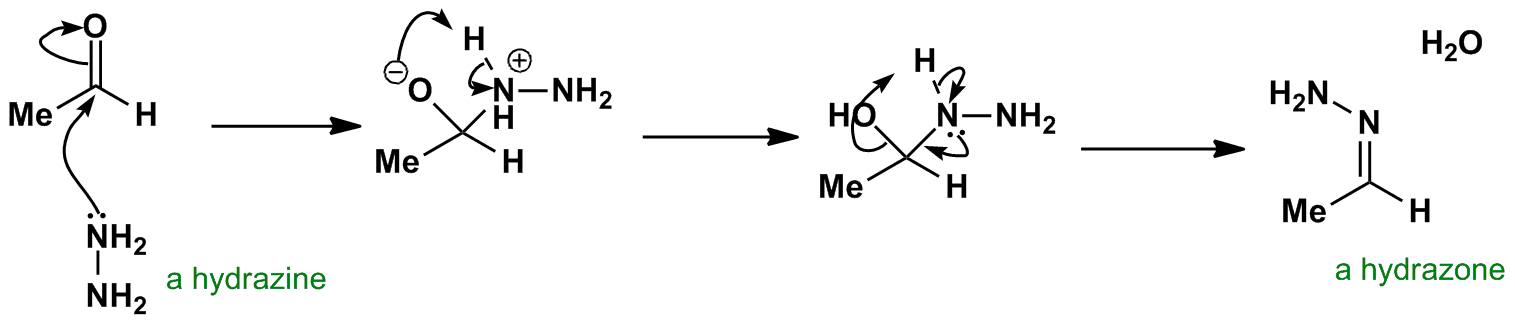
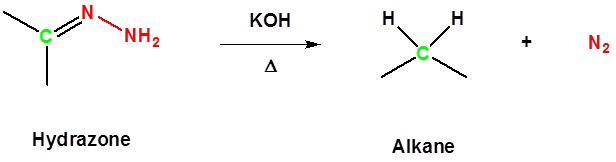
/chapter6/pages33and34/page33and34_files/WolffKishnermechanism.png)