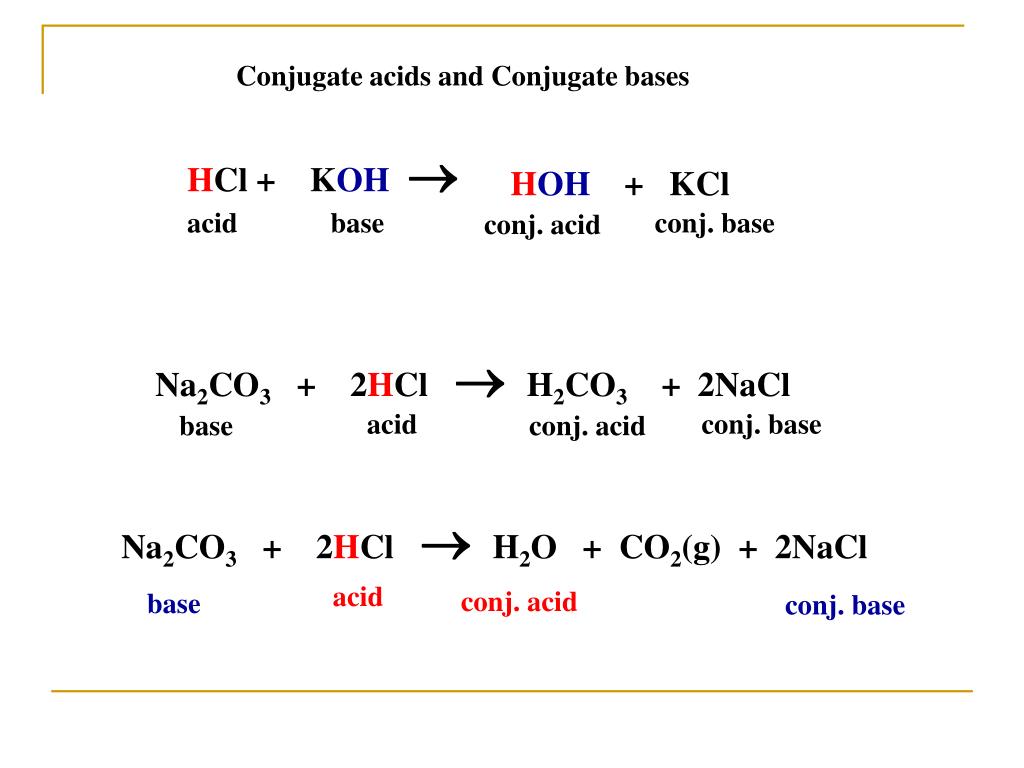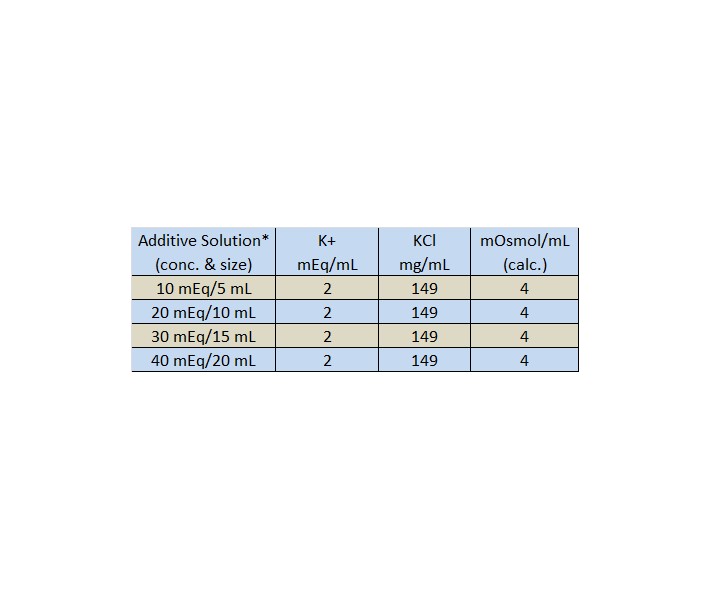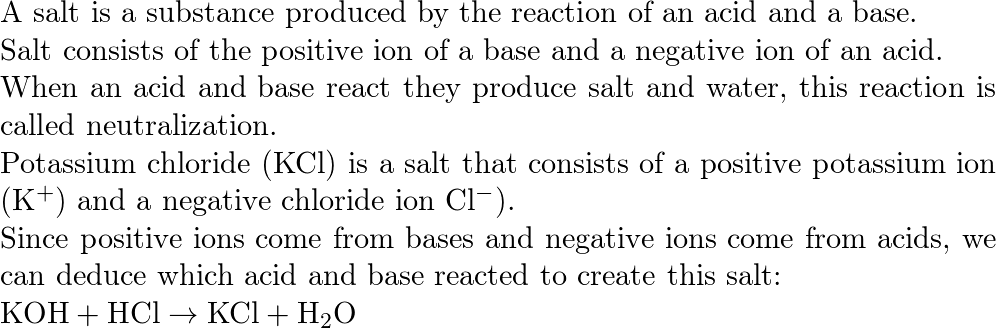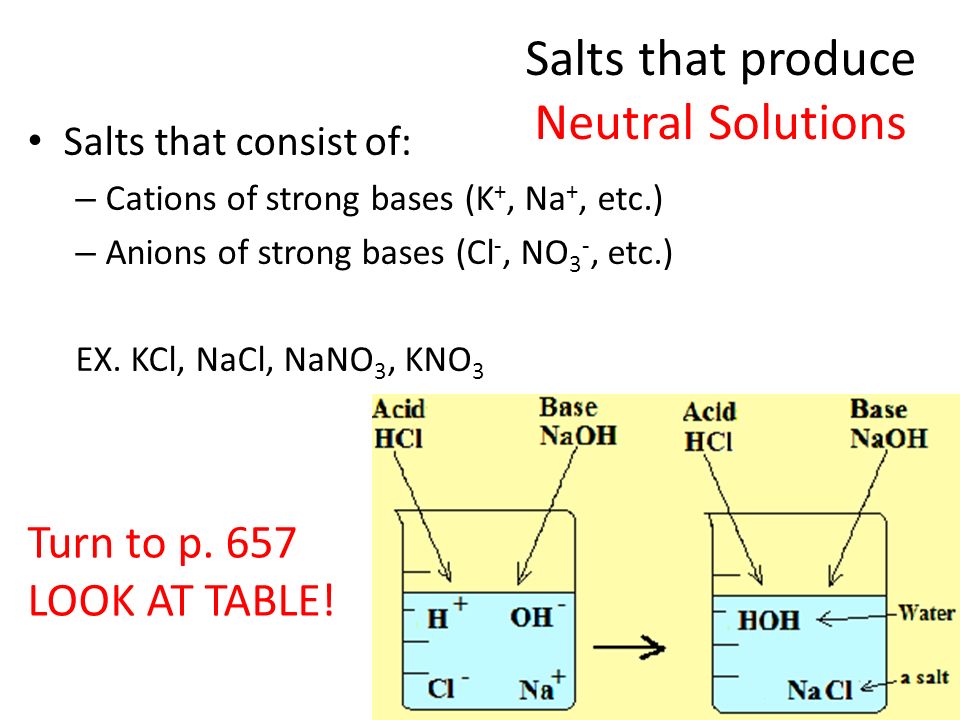
Salts as Acids and Bases Cations Sloshing Anions Sloshing Salt Ionic Compound Dissolves Ions Slosh Ions as Acids Ions as Bases Assuming that only one ion. - ppt download

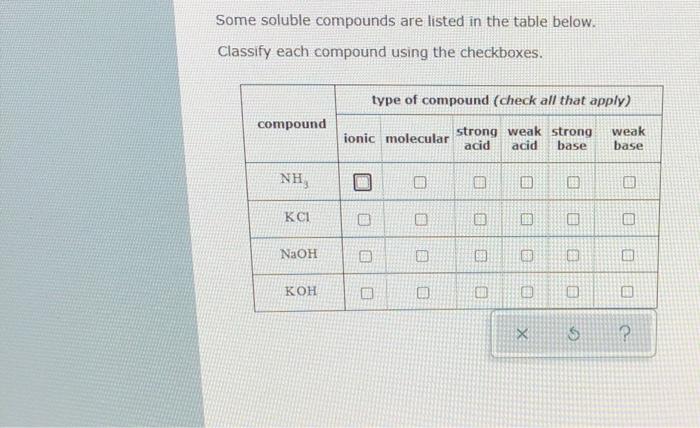
Acid-Base Properties of Salts. These salts simply dissociate in water: KCl(s) K + (aq) + Cl - (aq) - ppt download

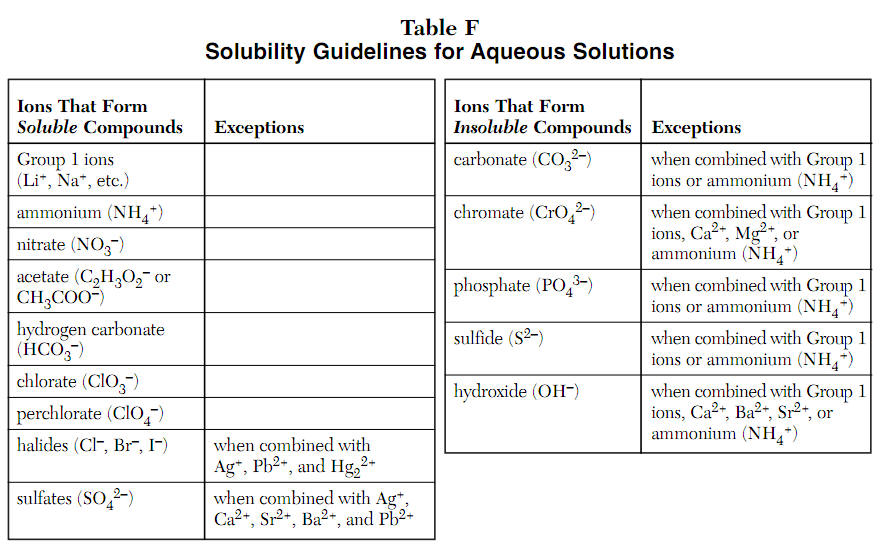
Acid and Base Production from 1.25 M NaCl, 0.2 M KCl, 0.008 M K 3 PO 4... | Download Scientific Diagram
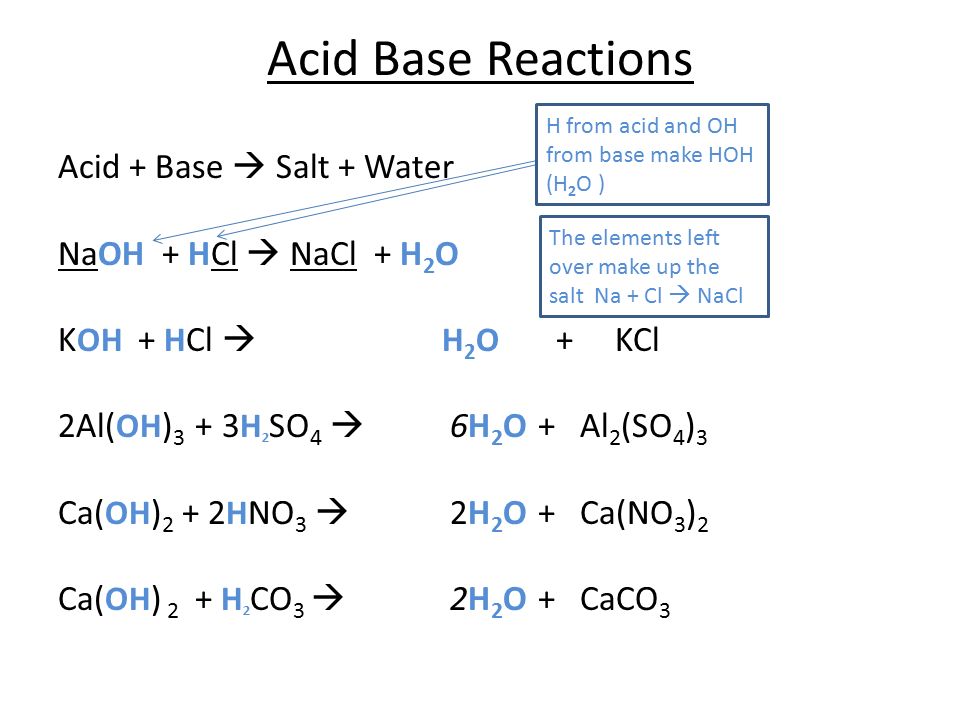
Acid and Base Chemistry. Some Properties of Acids þ Produce H + (as H 3 O + ) ions in water (the hydronium ion is a hydrogen ion attached to a water molecule) - ppt download