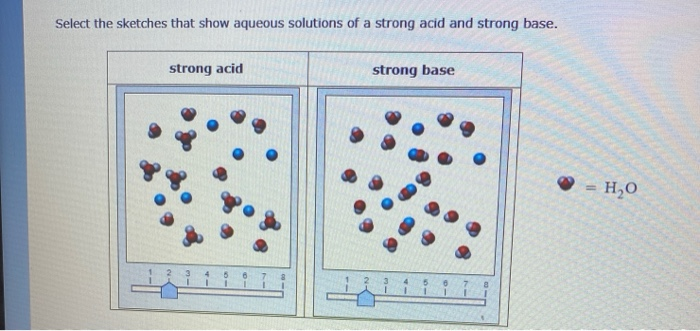
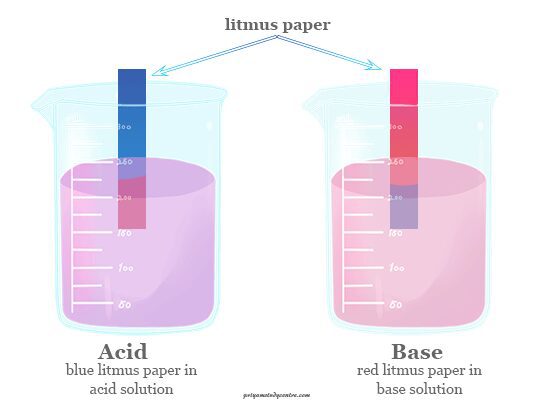
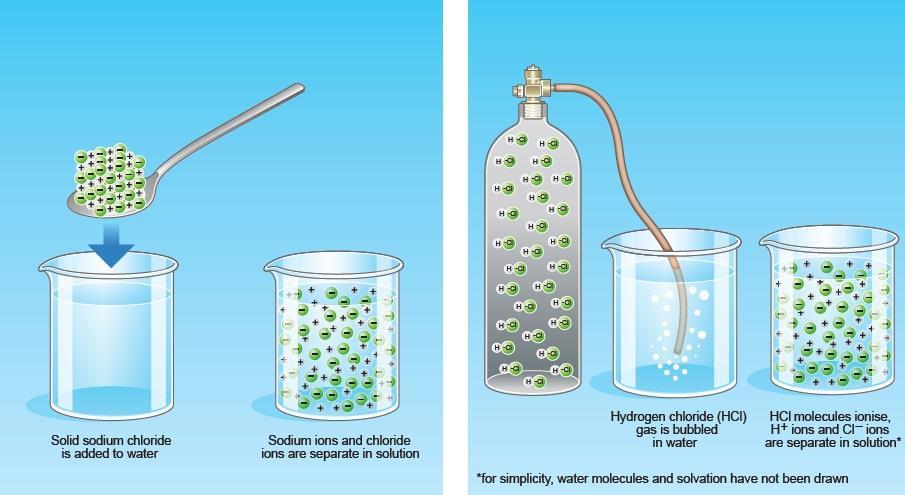
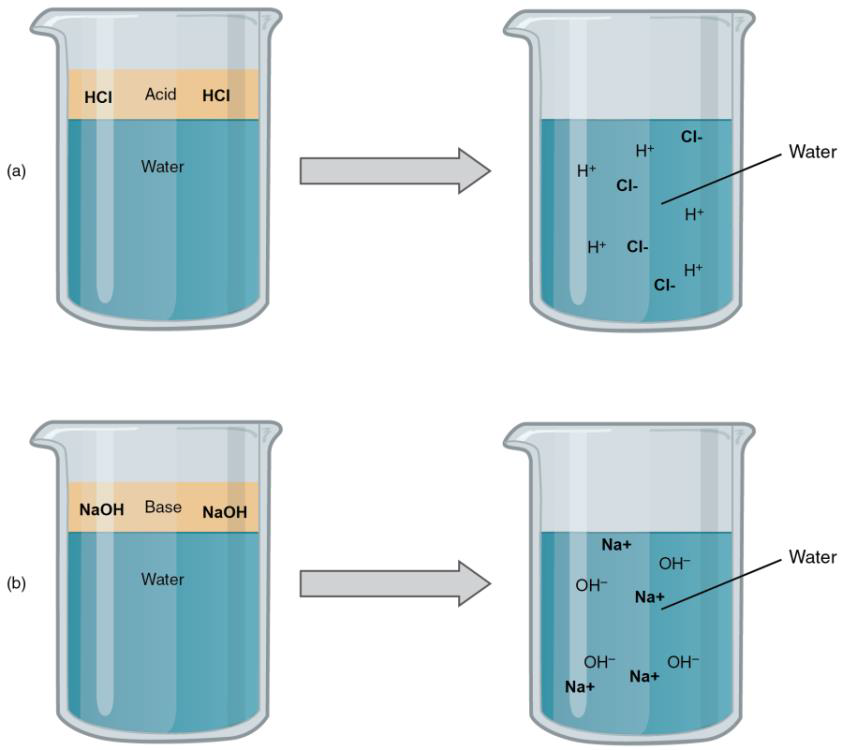
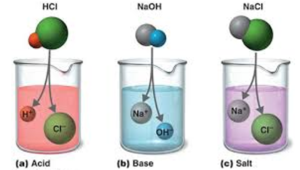
Sodium hydroxide (NaOH) is classified as a strong base. For every mole of sodium hydroxide added to a large volume of water, one mole of what ion enters the solution? | Socratic

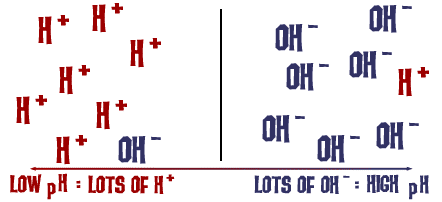
Acid-Base chemistry Acidity of blood (pH range of Heartburn (acid-reflux) – Tums, Rolaids, Milk of Magnesia; The Purple Pill , Nexium Acidity regulation. - ppt download



-in-water-01.jpg)